
Redesigning a certified bone growth stimulator to cut manufacturing costs and prepare it for the next generation of production

Healing a broken bone is all about bone growth. The Ossatec Bone Growth Stimulator promotes this process by stimulating the production of new cells. This makes the Bone Growth Stimulator ideal for both fresh fractures and fractures that heal slowly or not properly.
Client: Ossatec
Industry: MedTech
Service: PCB Design, Embedded Software, Back-End Development, Antenna Design

Client and Goals
Ossatec is a market leader in bone growth stimulation, a non-invasive therapy that uses electrical stimulation to accelerate fracture healing by up to 35%, reducing recovery times and in many cases eliminating the need for further surgery. When a new owner acquired the company, they needed a trusted engineering partner to take the existing Bone Growth Stimulator and make it fit for the future: lower manufacturing costs, reduced manual assembly effort, improved software reliability, and a clear path to scaled production, all without compromising the device's clinical efficacy or its regulatory standing.
Challenges
Redesigning a certified medical device is not like redesigning a consumer product. Every change to hardware or software has to be evaluated against existing regulatory approvals, and any modification that touches the device's therapeutic function requires careful validation. The pressure to reduce costs could not come at the expense of the clinical outcomes that made the product worth acquiring in the first place.
The existing hardware carried inefficiencies that had accumulated over the product's development history, component choices, manufacturing steps, and assembly processes that made sense at the time but added cost and labour at scale. The embedded software had reliability issues that needed resolving without introducing new failure modes. And throughout all of this, the new owner needed a partner who could coordinate across engineering, production, and assembly rather than hand off a design and walk away.
SPINNOV’s Solution
SPINNOV took on full responsibility for the redesign, working across hardware, software, and production preparation to deliver a device that met Ossatec's clinical standards while being meaningfully cheaper and simpler to manufacture. Specifically, we:
Redesigned the PCB to reduce component count, simplify the assembly process, and lower per-unit manufacturing costs
Optimised antenna design for reliable performance within the device's physical form factor
Resolved embedded software issues affecting device reliability and user experience
Streamlined the manufacturing and assembly process to reduce manual labour and improve consistency
Developed back-end infrastructure supporting device management and data handling
Maintained full alignment with healthcare regulatory requirements throughout every design decision
Delivered complete production-ready documentation covering hardware, software, and assembly


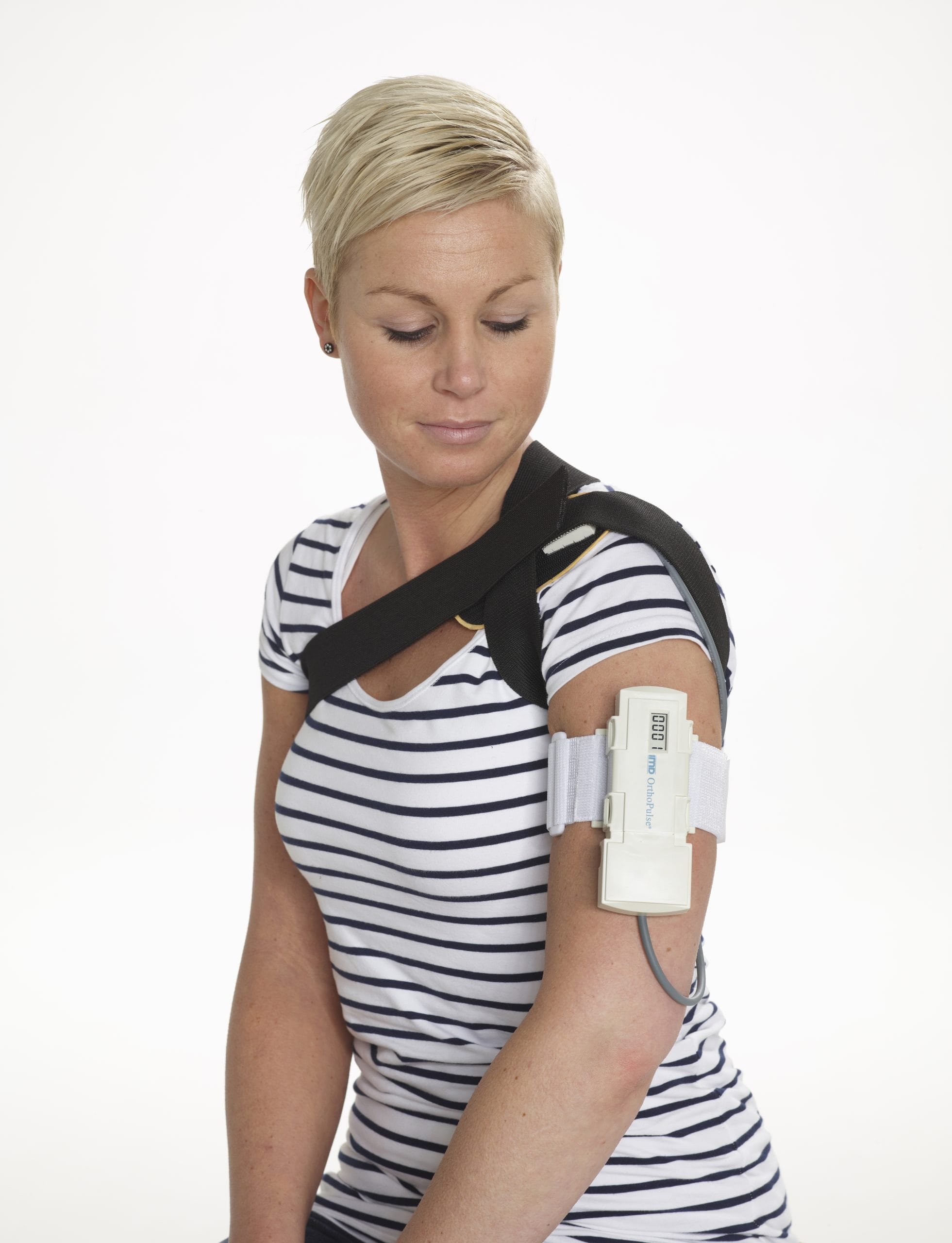

1 / Understanding the constraints before touching the design
Regulatory compliance as a design input, not an afterthought
The first thing we had to establish was what could change and what couldn't. A bone growth stimulator is a regulated medical device: its safety and efficacy claims rest on a body of clinical and technical evidence, and changes to the hardware or software can require regulatory re-evaluation if they affect how the device performs therapeutically.
We mapped the existing design carefully before proposing any modifications, identifying which components and subsystems were directly tied to the device's therapeutic function, which were purely manufacturing choices, and where the two overlapped. This gave us a clear picture of where we had freedom to optimise and where every change needed to be validated against the existing regulatory framework. It also meant the new owner had confidence that cost-reduction work wouldn't unexpectedly destabilise the product's market authorisation.
2 / Hardware redesign and cost reduction
Cutting cost without cutting corners
The PCB redesign focused on three areas: component rationalisation, assembly simplification, and supply chain resilience. Over a product's lifetime, component choices accumulate, parts selected for availability at a particular moment, workarounds for issues that have since been resolved, redundancies that made sense in early prototypes but add cost at volume. We reviewed the full bill of materials and eliminated what wasn't earning its place.
Assembly simplification mattered as much as component count. Manual assembly steps are slow, costly, and introduce variability, each one is an opportunity for error. We restructured the PCB layout and assembly sequence to reduce the number of manual operations required per unit, improving both throughput and consistency without requiring changes to the production facility itself.
The antenna redesign addressed a separate problem: ensuring the device's wireless performance was reliable and repeatable across production units, not dependent on precise manual placement or tuning steps that varied from unit to unit.
3 / Embedded software and reliability
Fixing what was fragile, not just what was broken
The embedded software issues we inherited fell into two categories: known bugs that affected specific use cases, and structural fragilities that hadn't caused visible problems yet but represented risk. We addressed both.
The known issues were resolved with fixes that were validated against the device's intended behaviour. The structural work was more careful, understanding why certain code paths were fragile, what assumptions they relied on, and how to make them robust without rewriting more than was necessary. In a regulated device, unnecessary change is risk. We kept our scope focused on what would genuinely improve reliability and left stable code alone.
The result was embedded software that behaved consistently across the device's operating conditions and gave the new owner a codebase they could understand, maintain, and extend.
